Services - Clinical Trial Report Preparation Services from Lucknow Uttar Pradesh India | ID - 3118976

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Clinical Trial Report Template (6) - TEMPLATES EXAMPLE | TEMPLATES EXAMPLE | Report template, Clinical trials, Template google

Trial Report Template (4) | PROFESSIONAL TEMPLATES in 2023 | Report template, Professional templates, Clinical trials

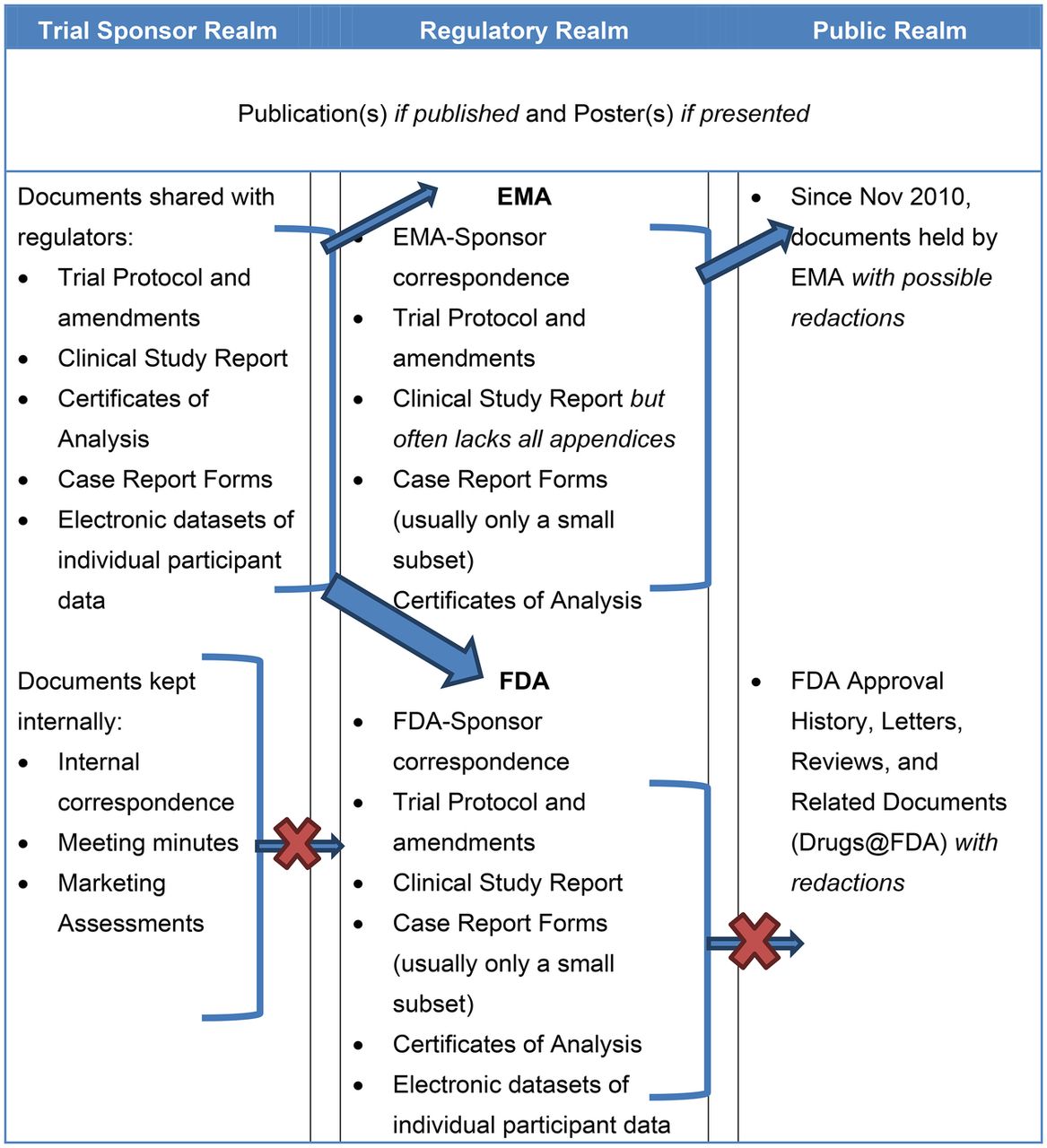
Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet

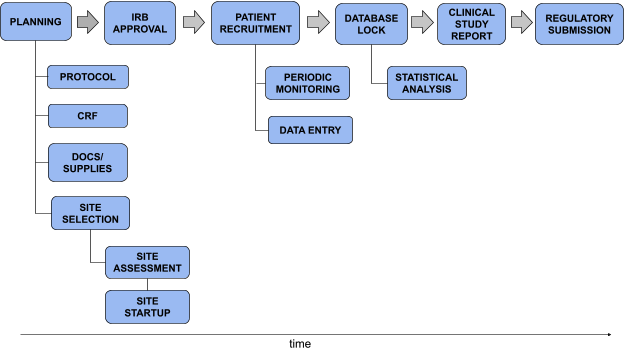
Construction of study sample comprising the 50 most recent clinical... | Download Scientific Diagram